Table of Contents
-The Properties of Water
-Properties of Water Lab
-Density Lab
-Plankton Lab
-The Properties of Water
-Properties of Water Lab
-Density Lab
-Plankton Lab

The Properties of Water
Water molecules are made of two hydrogen atoms bonded to a hydrogen one. Each end has a slight electric charge, making it a polar molecule. The hydrogen ends are positive and the oxygen ends are negative. This uneven distribution is called polarity. The positive hydrogen ends of water molecules attract the negative oxygen ends of others, so that water molecules form temporary, weak bonds called hydrogen bonds. The tendency of water to do this is called cohesion, which causes water molecules to be joined together as they move through the cells of organisms.
Surface tension, a special kind of cohesion, acts on the surface molecules of a liquid. It is the tightness across the surface caused by polar molecules pulling on each other downwards and sideways at the surface. This forms a tightly-pulled-together skin at the surface of the water. Some insects can walk across this.
Water also sticks to other things. This is called adhesion. Sometimes, it causes water to creep up surfaces. When it does this in a glass, it forms a curved surface called a meniscus. One particular kind of adhesion causes water to move up tubes like blood vessels and the insides of plants, to get all of an organism watered. This is called capillary action. The attraction between the water molecules and other materials causes it to climb up tubes against gravity.
A solution is a mixture caused by one substance dissolving another, caused by a solvent dissolving it. Water is the universal solvent because it dissolves more substances than any other. The positive charges attract the negatives parts molecules and the negative ones the positive parts, ripping the substance apart. Nonpolar substances like oil and wax do not dissolve in water, as they are hydrophobic and stay away from water.
Specific heat is the amount of heat needed to increase the temprature of 1 kg. of a substance by one degree celcius. Water has a high specific heat, 4,184 J/kg x C , so it takes a long time to heat p or cool down. This allows water ecosystems and your body to maintain stable temperatures.
Water molecules are made of two hydrogen atoms bonded to a hydrogen one. Each end has a slight electric charge, making it a polar molecule. The hydrogen ends are positive and the oxygen ends are negative. This uneven distribution is called polarity. The positive hydrogen ends of water molecules attract the negative oxygen ends of others, so that water molecules form temporary, weak bonds called hydrogen bonds. The tendency of water to do this is called cohesion, which causes water molecules to be joined together as they move through the cells of organisms.
Surface tension, a special kind of cohesion, acts on the surface molecules of a liquid. It is the tightness across the surface caused by polar molecules pulling on each other downwards and sideways at the surface. This forms a tightly-pulled-together skin at the surface of the water. Some insects can walk across this.
Water also sticks to other things. This is called adhesion. Sometimes, it causes water to creep up surfaces. When it does this in a glass, it forms a curved surface called a meniscus. One particular kind of adhesion causes water to move up tubes like blood vessels and the insides of plants, to get all of an organism watered. This is called capillary action. The attraction between the water molecules and other materials causes it to climb up tubes against gravity.
A solution is a mixture caused by one substance dissolving another, caused by a solvent dissolving it. Water is the universal solvent because it dissolves more substances than any other. The positive charges attract the negatives parts molecules and the negative ones the positive parts, ripping the substance apart. Nonpolar substances like oil and wax do not dissolve in water, as they are hydrophobic and stay away from water.
Specific heat is the amount of heat needed to increase the temprature of 1 kg. of a substance by one degree celcius. Water has a high specific heat, 4,184 J/kg x C , so it takes a long time to heat p or cool down. This allows water ecosystems and your body to maintain stable temperatures.
Properties of Water Lab
In this lab we explored some of the properties of water in several different stations. Here are pictures of a few of them.
In this lab we explored some of the properties of water in several different stations. Here are pictures of a few of them.

In this station we saw the meniscus of a glass of water. The adhesion, or sticking of water to other surfaces causes the surface of the water to curve, forming a curved surface. This same kind of adhesion helps water go up the roots of plants and into the branches.

This shows a certain kind of adhesion, called capillary action. It causes the water to travel from the first glass through the tube and into the other glass. This is the same type of adhesion I talked about in the earlier entry.
There are other stations we went to which I do not have pictures of. For instance, in one lab, we found out that water feels cooler than alcohol on your skin. This is because water absorbs heat before it evaporates, because it takes a long time to evaporate due to its high specific heat
In another, we studied another meniscus on a penny, formed by dropping water on it. The surface tension, caused by cohesion on the water surface, caused the water to keep from spilling over the top long after it went over the penny rim.
We also looked at what would happen to different liquids, one of which was water, when we put drops of them on wax paper. The water formed into a bead, while the other liquids spread out. The water did this because the cohesion, or attraction between the water molecules caused by polarity, made it stick together in the most efficient shape, a sphere, when it landed on the paper.
Finally, we looked at what would happen when we put paperclips on the surface of water. Most of them sunk, but some of them stayed on the surface because the surface tension held them up. When we put the dish soap in, the paperclips sunk to the bottom. This is because the dish soap disrupted the surface tension.
In another, we studied another meniscus on a penny, formed by dropping water on it. The surface tension, caused by cohesion on the water surface, caused the water to keep from spilling over the top long after it went over the penny rim.
We also looked at what would happen to different liquids, one of which was water, when we put drops of them on wax paper. The water formed into a bead, while the other liquids spread out. The water did this because the cohesion, or attraction between the water molecules caused by polarity, made it stick together in the most efficient shape, a sphere, when it landed on the paper.
Finally, we looked at what would happen when we put paperclips on the surface of water. Most of them sunk, but some of them stayed on the surface because the surface tension held them up. When we put the dish soap in, the paperclips sunk to the bottom. This is because the dish soap disrupted the surface tension.
Density Lab
In this lab, we used water, salt water, coloring and 2 beakers to look at the properties of density.
In this lab, we used water, salt water, coloring and 2 beakers to look at the properties of density.

First, we added blue food coloring and yellow food coloring to 2 different beakers filled with 250 ml. of water, with the yellow beaker containing warm, fresh water and the blue one containing cold, blue water with 20g of salt added.

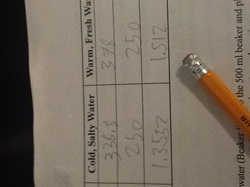
Then, we calculated the density of the water in the beakers, discovering that the warm, fresh water was somewhat less dense, at about 1.35 grams per cubic mililiter, than the cold, salty water, at 1.512 grams per cubic mililiter.

Finally, we emptied both beakers into a larger, 500 ml. beaker. Eventually the water mixed to become green, but for a while beforehand, the yellow water stayed at the top and the blue water at the bottom, with the salt at the absolute bottom of the beaker. This shows the principle of stratification, or how the ocean is divided into regions of greater and lesser density, separated by a thermocline. It often mixes because of wind up/down-welling and storms. This causes nutrients to go to the surface and oxygen to go to the bottom, making life possible in all of the ocean.
Bill Nye Buoyancy Video
Today we watched a video from Bill Nye about buoyancy and answered some questions about the video. Here are the questions and answers.
1. What does it mean to displace water?
To push water out of the way. It is caused when something enters a body of water and it makes the water level rise.
2. Explain why the clay ball sinks but the same size clay boat floats.
The shape of the boat allows it to stay on the water level, but the clay ball's shape makes it sink.
3. Things displace as much water as they weigh.
4. Who was Archimedes and what did he discover?
Archimedes was a Greek scientist who discovered displacement while taking a bath.
5. How do fish control their buoyancy?
Fish have a swim bladder that they use to regulate the amount of oxygen inside of them, and thus how much they float.
6. Define negatively, positively, and neutrally buoyant.
If something is negatively buoyant, it sinks.
If something is positively buoyant, it floats.
If something is neutrally buoyant, it stays in place and neither sinks nor floats.
7. Explain how hot air balloons work.
The less dense hot air in the balloon is lighter than the air around it, so the balloon is positively buoyant and it floats.
Today we watched a video from Bill Nye about buoyancy and answered some questions about the video. Here are the questions and answers.
1. What does it mean to displace water?
To push water out of the way. It is caused when something enters a body of water and it makes the water level rise.
2. Explain why the clay ball sinks but the same size clay boat floats.
The shape of the boat allows it to stay on the water level, but the clay ball's shape makes it sink.
3. Things displace as much water as they weigh.
4. Who was Archimedes and what did he discover?
Archimedes was a Greek scientist who discovered displacement while taking a bath.
5. How do fish control their buoyancy?
Fish have a swim bladder that they use to regulate the amount of oxygen inside of them, and thus how much they float.
6. Define negatively, positively, and neutrally buoyant.
If something is negatively buoyant, it sinks.
If something is positively buoyant, it floats.
If something is neutrally buoyant, it stays in place and neither sinks nor floats.
7. Explain how hot air balloons work.
The less dense hot air in the balloon is lighter than the air around it, so the balloon is positively buoyant and it floats.

Plankton Lab
In this lab, we were looking at how plankton move and function in the water. First, we were taught about what plankton are, what different types there were, and how and where they moved and got their food. Then we performed an experiment to help discover exactly how these plankton worked. We made plankton models and timed how long it took for them to sink.
In this lab, we were looking at how plankton move and function in the water. First, we were taught about what plankton are, what different types there were, and how and where they moved and got their food. Then we performed an experiment to help discover exactly how these plankton worked. We made plankton models and timed how long it took for them to sink.
Phytoplankton-Use the sun to photosynthesize.
Photic zone-The well lit area of the ocean that phytoplankton stay in to get light.
Zooplankton-Larger plankton that eat phytoplankton. They need to go up to the photic zone to find them, but since they are drifters and not good swimmers they need special adaptions to remain afloat in the photic zone
Dinoflagellates-A type of phytopplankton that use flagella, long tails, to stay afloat.
Ciliates-Plankton that use hair-like extensions called cilia to float.
Some plankton form chains to float.
Vacuoles-Compartments that some plankton use to stay afloat by providing buoyancy.
Benthic-Plankton found on the bottom of a body of water on rocks and sediment.
Some plankton attatch themselves to vegetation in water.
Photic zone-The well lit area of the ocean that phytoplankton stay in to get light.
Zooplankton-Larger plankton that eat phytoplankton. They need to go up to the photic zone to find them, but since they are drifters and not good swimmers they need special adaptions to remain afloat in the photic zone
Dinoflagellates-A type of phytopplankton that use flagella, long tails, to stay afloat.
Ciliates-Plankton that use hair-like extensions called cilia to float.
Some plankton form chains to float.
Vacuoles-Compartments that some plankton use to stay afloat by providing buoyancy.
Benthic-Plankton found on the bottom of a body of water on rocks and sediment.
Some plankton attatch themselves to vegetation in water.
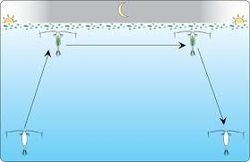
Most zooplankton go through a diurnal migration where they go to deeper water during the day and return to the top at night. They do this to conserve energy and avoid predation.
Diatoms use a raphe, or a slit along the side of their cell where tiny microfribils protrude, to move over a surface.
All these are adaptions, physical or behavioral traits allowing an organism to better survive.
Surface area-total area of all faces and curved surfaces of a solid object.
Density- mess per unit of volume, measure of relative heaviness.
Micron-1/1000th of a millimeter. Used for measuring plankton size.
Diatoms use a raphe, or a slit along the side of their cell where tiny microfribils protrude, to move over a surface.
All these are adaptions, physical or behavioral traits allowing an organism to better survive.
Surface area-total area of all faces and curved surfaces of a solid object.
Density- mess per unit of volume, measure of relative heaviness.
Micron-1/1000th of a millimeter. Used for measuring plankton size.
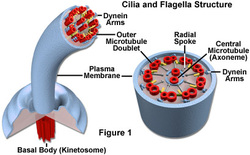
All the appendages on a plankton have a purpose. Flagella, legs/bristles, cilia and antennae are used for motion. Flagella, cilia, eyes and translucent skin are used to avoid predators. Legs/bristles, antennae and eyes are used to catch phytoplankton play. Varioles/swim bladders, wings, swim bladders and translucency are used to stay afloat.
Varioles/swim bladders are used for moving up and down by controlling buoyancy.
1. Plankton are small, mostly microscopic organisms that drift in bodies of water.
2. Zoolankton eat smaller plankton and phytoplankton, while phytoplankton photosynthesize.
3. Plankton are the basis of most underwater food chains and are very important.
4. Phytoplankton want to stay at the top, the photic zone, because that is where they can get enough sun to photosynthesize, and zooplankton want to go there only during the night to eat phytoplankton.
5/6. A plankton could stay in the photic zone with swim bladders, appendages like wings, or thin skin or other things to reduce weight.
Varioles/swim bladders are used for moving up and down by controlling buoyancy.
1. Plankton are small, mostly microscopic organisms that drift in bodies of water.
2. Zoolankton eat smaller plankton and phytoplankton, while phytoplankton photosynthesize.
3. Plankton are the basis of most underwater food chains and are very important.
4. Phytoplankton want to stay at the top, the photic zone, because that is where they can get enough sun to photosynthesize, and zooplankton want to go there only during the night to eat phytoplankton.
5/6. A plankton could stay in the photic zone with swim bladders, appendages like wings, or thin skin or other things to reduce weight.

This is me and Christian's plankton, Sheldon. It is named that because it is Plankton from Spongebob's real name. To keep it from sinking fast, we added a buoyancy chamber consisting of a straw in the middle. We put clay on it so that it wouldn't float. Finally, we put wires and cotton on it as wings to further slow down its sinking.
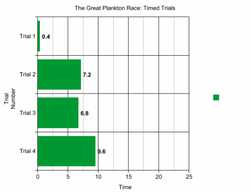
Results/Conclusion
Our average time to get to the bottom was 6 seconds. This was shorter than we expected, but above average compared to the rest of our classmates. My observations included that that the plankton that stayed the most were the ones that had the greatest balance and the most appendages to stop sinking. These are what help plankton that move in the water column, and what the best planktons in the class used. They looked similar to ours but were more complex. Saltwater is less dense than freshwater, and thus the sinking time would be shorter for our plankton. This would also be true for cold water, less dense, and hot water, more dense. Other than buoyancy, plankton evolution is also influenced by predation avoidance and prey catching. We learned that plankton have to adapt well to get to survive, thus evolution is constantly changing them
Our average time to get to the bottom was 6 seconds. This was shorter than we expected, but above average compared to the rest of our classmates. My observations included that that the plankton that stayed the most were the ones that had the greatest balance and the most appendages to stop sinking. These are what help plankton that move in the water column, and what the best planktons in the class used. They looked similar to ours but were more complex. Saltwater is less dense than freshwater, and thus the sinking time would be shorter for our plankton. This would also be true for cold water, less dense, and hot water, more dense. Other than buoyancy, plankton evolution is also influenced by predation avoidance and prey catching. We learned that plankton have to adapt well to get to survive, thus evolution is constantly changing them
